Electrochemical Corrosion
How it works, how to control it

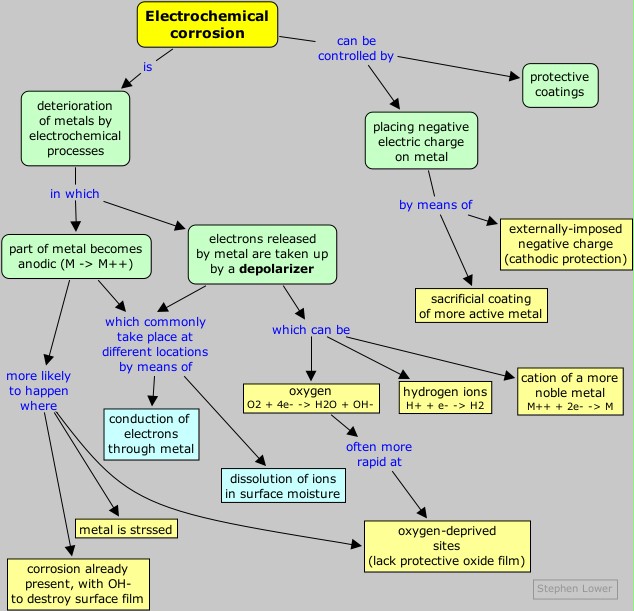
Corrosion can be defined as the deterioration of materials by chemical processes. Of these, the most important by far is electrochemical corrosion of metals, in which the oxidation process M → Mn+ + n en– is facilitated by the presence of a suitable electron acceptor, sometimes referred to in corrosion science as a depolarizer.
In a sense, corrosion can be viewed as the spontaneous return of metals to their ores; the huge quantities of energy that were consumed in mining, refining, and manufacturing metals into useful objects is dissipated by a variety of different routes.
The economic aspects of corrosion are far greater than most people realize; according to a 2001 report, the cost of corrosion in the U.S. alone was $276 billion per year. Of this, about $121 billion was spent to control corrosion, leaving the difference of $155 billion as the net loss to the economy. Utilities, especially drinking water and sewer systems, suffer the largest economic impact, with motor vehicles and transportation being a close second.
The special characteristic of most corrosion processes is that the oxidation and reduction steps occur at separate locations on the metal. This is possible because metals are conductive, so the electrons can flow through the metal from the anodic to the cathodic regions. The presence of water is necessary in order to transport ions to and from the metal, but a thin film of adsorbed moisture can be sufficient.
A corrosion system can be regarded as a short-circuited electrochemical cell in which the anodic process is something like
Fe(s) → Fe2+(aq) + 2 e–
and the cathodic steps can be any of
O2 + 2 H2O + 4e– → 4 OH–
H+ + e– → ½ H2(g)
M2+ + 2 e– → M(s)
where M is a metal.
Which parts of the metal serve as anodes and cathodes can depend on many factors, as can be seen from the irregular corrosion patterns that are commonly observed. Atoms in regions that have undergone stress, as might be produced by forming or machining, often tend to have higher free energies, and thus tend to become anodic.
[link]
If one part of a metallic object is protected from the atmosphere so that there is insufficient O2 to build or maintain the oxide film, this "protected" region will often be the site at which corrosion is most active. The fact that such sites are usually hidden from view accounts for much of the difficulty in detecting and controlling corrosion.
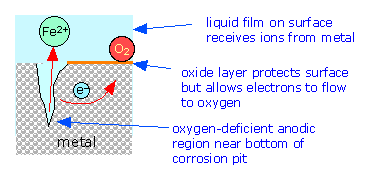
The tendency of oxygen-deprived locations to become anodic is the cause of many commonly-observed patterns of corrosion
In contrast to anodic sites, which tend to be localized to specific regions of the surface, the cathodic part of the process can occur almost anywhere. Because metallic oxides are usually semiconductors, most oxide coatings do not inhibit the flow of electrons to the surface, so almost any region that is exposed to O2 or to some other electron acceptor can act as a cathode.
Rusted-out cars and bathroom stains
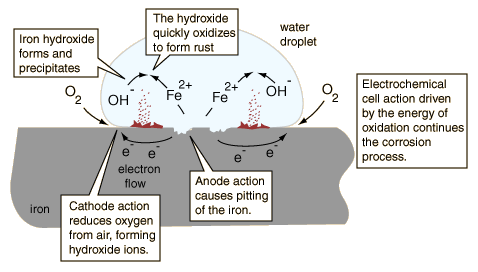
 Anyone who has owned an older car has seen corrosion occur at joints between body parts and under paint films. You will also have noticed that once corrosion starts, it tends to feed on itself. One reason for this is that one of the products of the O2 reduction reaction is hydroxide ion. The high pH produced in these cathodic regions tends to destroy the protective oxide film, and may even soften or weaken paint films, so that these sites can become anodic. The greater supply of electrons promotes more intense cathodic action, which spawns even more anodic sites, and so on.
Anyone who has owned an older car has seen corrosion occur at joints between body parts and under paint films. You will also have noticed that once corrosion starts, it tends to feed on itself. One reason for this is that one of the products of the O2 reduction reaction is hydroxide ion. The high pH produced in these cathodic regions tends to destroy the protective oxide film, and may even soften or weaken paint films, so that these sites can become anodic. The greater supply of electrons promotes more intense cathodic action, which spawns even more anodic sites, and so on.
A very common cause of corrosion is having two dissimilar metals in contact, as might occur near a fastener or at a weld joint. Moisture collects at the junction point, acting as an electrolyte and forming a cell in which the two metals serve as electrodes. Moisture and conductive salts on the outside surfaces provide an external conductive path, effectively short-circuiting the cell and producing very rapid corrosion; this is why cars rust out so quickly in places where salt is placed on roads to melt ice.
Dissimilar-metal corrosion can occur even if the two metals are not initially in direct contact. For example, in homes where copper tubing is used for plumbing, there is always a small amount of dissolved Cu2+ in the water. When this water encounters steel piping or a chrome-plated bathroom sink drain, the more-noble copper will plate out on the other metal, producing a new metals-in-contact corrosion cell. In the case of chrome bathroom sink fittings, this leads to the formation of Cr3+ salts which precipitate as greenish stains.
Since both the cathodic and anodic steps must take place for corrosion to occur, prevention of either one will stop corrosion. The most obvious strategy is to stop both processes by coating the object with a paint or other protective coating. Even if this is done, there are likely to be places where the coating is broken or does not penetrate, particularly if there are holes or screw threads.
A more sophisticated approach is to apply a slight negative charge to the metal, thus making it more difficult for the reaction M → M2+ + 2 e– to take place.
How sacrificial coatings control corrosion
One way of supplying this negative charge is to apply a coating of a more active metal. Thus a very common way of protecting steel from corrosion is to coat it with a thin layer of zinc; this process is known as galvanizing.The zinc coating, being less noble than iron, tends to corrode selectively. Dissolution of this sacrificial coating leaves behind electrons which concentrate in the iron, making it cathodic and thus inhibiting its dissolution.
Cathodic protection for corrosion control
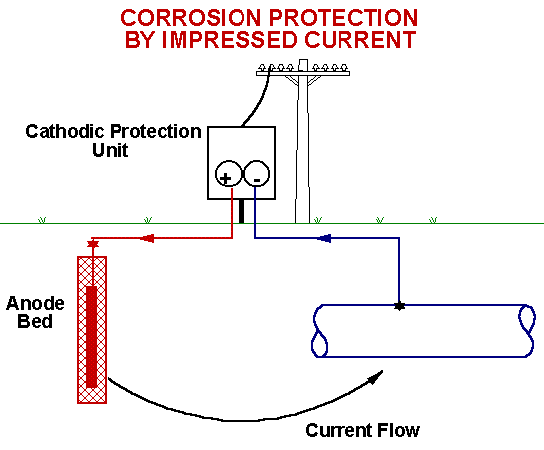 A more sophisticated strategy is to maintain a continual negative
electrical charge on a metal, so that its dissolution as positive ions
is inhibited. Since the entire surface is forced into the cathodic
condition, this method is known as cathodic protection.
The source of electrons can be an external direct current power supply
(commonly used to protect oil pipelines and other buried structures),
or it can be the corrosion of another, simply a piece of amore active metal such as zinc or aluminum buried in the ground nearby.
A more sophisticated strategy is to maintain a continual negative
electrical charge on a metal, so that its dissolution as positive ions
is inhibited. Since the entire surface is forced into the cathodic
condition, this method is known as cathodic protection.
The source of electrons can be an external direct current power supply
(commonly used to protect oil pipelines and other buried structures),
or it can be the corrosion of another, simply a piece of amore active metal such as zinc or aluminum buried in the ground nearby.
Corrosion of a nail
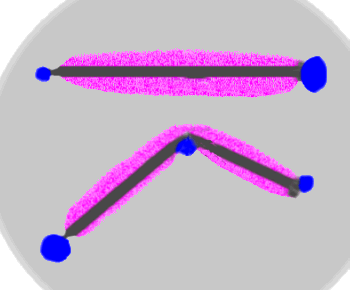
The nails are immersed in agar which forms a moist solid gel. The agar also contains phenolphthalein and hexacyanoiron(III) Fe(CN6)– which forms a deep blue color ("prussian blue") in the presence of Fe2+. The blue colors are clearly associated with those parts of the nail that have been stressed, thus faciliting the anodic release of Fe2+ from the metal. The the pink color shows the cathodic regions that have been made alkaline by the reaction O2 + 2 H2O + 4e– → 4 OH– This clearly shows the separation between the anodic and cathodic processes in corrosion.
Interior of a water distribution main
 If you live in the older part of a city where the mains are 50-100 years old, the water you drink may well have passed through a pipe in this condition! Severe corrosion like this is more common in areas where the water is acidic. Such water comes from mountain snowmelt and runoff, and usually acquires its acidity from dissolved atmospheric carbon dioxide.
If you live in the older part of a city where the mains are 50-100 years old, the water you drink may well have passed through a pipe in this condition! Severe corrosion like this is more common in areas where the water is acidic. Such water comes from mountain snowmelt and runoff, and usually acquires its acidity from dissolved atmospheric carbon dioxide.
Waters from rivers, lakes, and especially groundwaters from wells have usually been in sufficiently long contact with carbonate-containing sediments to have been neutralized. Water-works administrators like to make the water slightly alkaline and slightly supersaturated in calcium carbonate in order to maintain a thin coating of solid carbonate on the interior of the pipe which acts to protect it from corrosion.
Steel reinforcing bars embedded in concrete

All large concrete structures contain steel reinforcing bars ("rebars") that help ensure structural integrity under varying load conditions and especially during earthquakes. Intrusion of water, even in the form of fog or mists, can lead to serious corrosion damage, as seen in this picture of this column which supports a highway overpass.\\
Corrosion at metallic joints
 The picture shows two steel structural members joined by cast iron flanges which have been bolted together. For some reason, one of the pieces has become more anodic than the other, leading to extensive corrosion of the upper part.
The picture shows two steel structural members joined by cast iron flanges which have been bolted together. For some reason, one of the pieces has become more anodic than the other, leading to extensive corrosion of the upper part.
Corrosion mediated by soil bacteria
 This gas pipe was buried in a red soil that contained iron pyrites (FeS.) The bacterium thiobacillus ferrooxidans derives its energy by oxidizing Fe2+ to the more soluble Fe3+, transferring the electrons to O2. It also oxidizes the sulfur, producing sulfuric acid. The resulting chemical cocktail has eaten a hole into the pipe.
This gas pipe was buried in a red soil that contained iron pyrites (FeS.) The bacterium thiobacillus ferrooxidans derives its energy by oxidizing Fe2+ to the more soluble Fe3+, transferring the electrons to O2. It also oxidizes the sulfur, producing sulfuric acid. The resulting chemical cocktail has eaten a hole into the pipe.
These galvanized bolts were used to join wooden beams together. Subsequent movement of the beams due to varying load conditions abraded the zinc coating. A lack of oxygen near the centers of the bolts also likely contributed to the corrosion by preventing the formation of a protective oxide film.
Corrosion by lack of oxygen

Pitting corrosion of a surface

Pitting corrosion tends to produce narrow but deep holes ("pits") in a surface that can be difficult to detect, especially when they get covered up by corrosion products. Once a pit develops, it tends to dig itself deeper owing to the slower diffusion of oxygen into the narrow bottom of the pit.

Stainless steel is not entirely immune to corrosion
 Steel alloys containing 13-26 weight-percent chromium develop a thin, highly adherent surface layer of Cr2O3 when exposed to air, thereby becoming passivated and resistant to corrosion. But in order to retain this resistance, oxygen must continue to be available to the surface.
Steel alloys containing 13-26 weight-percent chromium develop a thin, highly adherent surface layer of Cr2O3 when exposed to air, thereby becoming passivated and resistant to corrosion. But in order to retain this resistance, oxygen must continue to be available to the surface.
If this condition is not maintained, the passivating film can break down, as has happened to this flange which has suffered extensive pitting corrosion. Chloride ions can also disrupt the protective film, although there are special grades of stainless steel available for marine use that are more resistant to this effect.
 Special care must be taken when stainless steels are joined to other structures. Over-tightening the nuts on this bracket could have damaged the Cr2O3 film; the use of non-SS machine screws and nuts would guarantee ordinary galvanic corrosion.
Special care must be taken when stainless steels are joined to other structures. Over-tightening the nuts on this bracket could have damaged the Cr2O3 film; the use of non-SS machine screws and nuts would guarantee ordinary galvanic corrosion.
Make sure you thoroughly understand the following essential concepts that have been presented above.
- Electrochemical corrosion of metals occurs when electrons from atoms at the surface of the metal are transferred to a suitable electron acceptor or depolarizer. Water must be present to serve as a medium for the transport of ions.
- The most common depolarizers are oxygen, acids, and the cations of less active metals.
- Because the electrons flow through the metallic object itself, the anodic and cathodic regions (the two halves of the electrochemical cell) can be at widely separated locations.
- Anodic regions tend to develop at locations where the metal is stressed or is protected from oxygen.
- Contact with a different kind of metal, either direct or indirect, can lead to corrosion of the more active one.
- Corrosion of steel can be inhibited by galvanizing, that is, by coating it with zinc, a more active metal whose dissolution leaves a negative charge on the metal which inhibits the further dissolution of Fe2+.
- Cathodic protection using an external voltage source is widely used to protect underground structures such as tanks, pipelines and piers. The source can be a sacrificial anode of zinc or aluminum, or a line-operated or photovoltaic power supply.