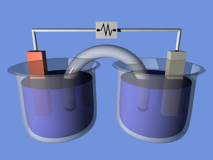
The connection between chemistry and electricity is a very old one, going back to Alessandro Volta's discovery, in 1793, that electricity could be produced by placing two dissimilar metals on opposite sides of a moistened paper.
 |
“I have the pleasure of communicating to you, Sir, and through you to the Royal Society, some striking results at which I have arrived in pursuing my experiments on the electricity excited by the simple mutual contact of metals of different sorts...” |  |

In 1800, Nicholson and Carlisle, using Volta's primitive battery as a source, showed that an electric current could decompose water into oxygen and hydrogen.
This was the first proof that water is composed of hydrogen and oxygen.
This was surely one of the most significant experiments in the history of chemistry, for it implied that the atoms of hydrogen and oxygen were associated with positive and negative electric charges, which must be the source of the bonding forces between them.
By 1812, the Swedish chemist Berzelius could propose that all atoms are electrified, hydrogen and the metals being positive, the nonmetals negative. In electrolysis, the applied voltage was thought to overpower the attraction between these opposite charges, pulling the electrified atoms apart in the form of ions (named by Berzelius from the Greek for "travelers"). It would be almost exactly a hundred years later before the shared electron pair theory of G.N. Lewis could offer a significant improvement over this view of chemical bonding.
Meanwhile the use of electricity as a means of bringing about chemical change continued to play a central role in the development of chemistry. Humphrey Davey prepared the first elemental sodium by electrolysis of a sodium hydroxide melt.
 It was left to Davey's former assistant, Michael Faraday, to show that there is a direct relation between the amount of electric charge passed through the solution and the quantity of electrolysis products. James Clerk Maxwell immediately saw this as evidence for the "molecule of electricity", but the world would not be receptive to the concept of the electron until the end of the century.
It was left to Davey's former assistant, Michael Faraday, to show that there is a direct relation between the amount of electric charge passed through the solution and the quantity of electrolysis products. James Clerk Maxwell immediately saw this as evidence for the "molecule of electricity", but the world would not be receptive to the concept of the electron until the end of the century.
Electron-transfer reactions: oxidation and reduction
We generally think of a "chemical reaction" as a process in which the atoms in one or more reactants undergo some kind of a re-arrangment into new substances, the products of the reaction. There are, however, many reactions in which it is convenient to regard electrons as participants in the transformation of reactants into products. Reactions of this kind are known generally as electron-transfer reactions, or more commonly as oxidation-reduction ("redox") reactions.
It would be incorrect to view electrons themselves as reactants or products because they are not "substances" that can exist independently of the atoms or molecules to which they are always attached.
The terms "oxidation" and "reduction" originally described certain types of reactions that were known long before the role of electrons in atoms and the chemical bonds that link them together became clear in the 1920s. Once it became known that these same kinds of reactions involved the loss and gain of electrons, the definitions of the two words were generalized to reflect this fact.
Oxidation
As its name implies, "oxidation" originally referred to the combination of a substance with oxygen, a process that, when it takes place rapidly with the release of sufficient heat to produce a flame, was (and still is) known as combustion. An example would be the combustion of elemental sulfur:
S + O2 → SO2(1-1)
As we will see later, this kind of reaction can also be regarded as one in which electrons are transferred from one reactant (sulfur) to the other (oxygen). In other words, the sulfur having lost electrons, has undergone oxidation: it has been oxidized.
Now consider another reaction that is often demonstrated in class or carried out in the lab: when a piece of zinc metal is immersed in an aqueous solution of copper(II) sulfate, the surface of the zinc is rapidly covered with a black coating of metallic copper:
Zn + Cu2+ → Cu + Zn2+(1-2)
The transfer of the electric charge from the copper(II) ion to zinc means that two electrons have been transferred from the zinc atom to the copper. In this sense, the reactions 1 and 2 are functionally similar, in that one of the reactants (sulfur in 1, and zinc in 2) has lost electrons, and, by definition, has undergone oxidation.
Oxidation refers to the loss of electrons. Important!
Reduction
This term originally referred to the loss in weight of a metallic ore when it is "reduced" to a metal. For example, when the ore mercuric oxide is heated, the oxygen is driven off as O2, liberating the pure metal:
HgO → Hg + ½ O2(1-3)
Rewriting this equation with Lewis dot notation showing the bonding electron pairs (omitting the non-bonding electrons on the oxygens) shows that in HgO, the Hg atom effectively "owns" only one of the two electrons it shares with oxygen, so it gains an electron in the process, which corresponds to the modern generalized definition of "reduction":
Hg:O → Hg: + ½ O::O(1-4)
Reduction refers to the gain of electrons. Important!
Because loss of electrons by one substance implies the gain of electrons by another, oxidation and reduction always occur together, in a transactional process known as oxidation-reduction, commonly abbreviated as "redox".
Electrochemistry is the study of reactions in which charged particles (ions or electrons) cross the interface between two phases of matter, typically a metallic phase (the electrode) and a conductive solution, or electrolyte. A process of this kind can always be represented as an electrode reaction in which one or more electrons appear explicity in the reaction equation, either as reactants ("oxidation") or as products ("reduction"):
| reduction: | Cu2+(aq) + 2e– → Cu(s) |
| oxidation: | Cu(s)→ Cu2+(.aq) + 2e– |
The electroneutrality principle
Nature seems to strongly discourage any process that would lead to an excess of positive or negative charge in bulk matter. This is the essence of the electroneutrality principle, which states that bulk matter (a piece of metal, or a liquid) cannot sustain more than a minute electric charge.
There is nothing mysterious about this prohibition; it is a simple consequence of the thermodynamic work required to separate opposite charges, or to bring like charges into closer contact. The additional work raises the free energy change of the process, making it less spontaneous.
When a metal such as iron, copper, or zinc is immersed in an electrolyte, a small quantity of metal cations will be released into the solution.
For example, if the metal is zinc and the solution contains ZnNO3,
the process can be described by
Zn(s) → Zn+(aq)
You will notice that no electrons are shown here. Where did they go?
A metal can be thought of as an assembly of positive ions immersed in a fluid-like sea of electrons, whose mobility is responsible for the typical metallic properties such as electrical and heat conductivity. As Zn+ ions leave the metal and attach themselves to water molecules, the electron left behind cause the metal to assume a negative charge. As this negative charge builds up, it makes it energetically more difficult for the metal to release cations into the solution. Similarly, the Zn+ ions released into the solution create a positive charge that soon begins to inhibit their further dissolution. As a result, the reaction quickly grinds to a halt before any chemically significant concentration of Zn2+ ions ions appears in the solution.
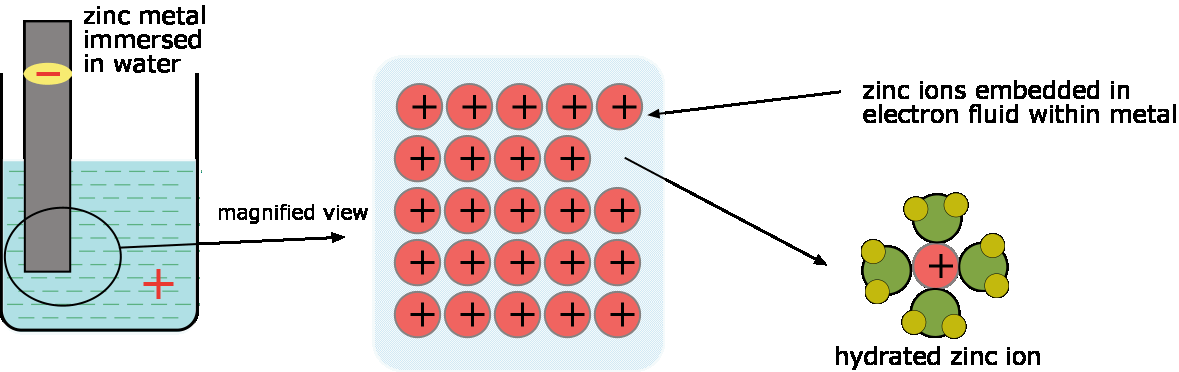
The concentration of hydrated ions Zn+(aq) in the water will typically be around 10–10 M, which for most practical purposes is neglibible.
Giving the electrons some place to go
The only way we can get the oxidation of the metal to continue is to couple it with some other process that restores electroneutrality to both the metal and the liquid. A simple way to accomplish this would be to immerse the zinc in a solution of copper sulfate instead of pure water. As you will recall if you have seen this commonly-performed experiment carried out, the zinc metal quickly becomes covered with a black coating of finely-divided metallic copper. The reaction is a simple oxidation-reduction process, a transfer of two electrons from the zinc to the copper:
Zn(s) → Zn2+ + 2e– Cu2+ + 2e– → Cu(s)
The dissolution of the zinc is no longer inhibited by a buildup of negative charge in the metal, because the excess electrons are removed from the zinc by copper ions that come into contact with it. At the same time, the solution remains electrically neutral, since for each Zn2+ ion introduced to the solution, one Cu2+ ion is removed. The net reaction
Zn(s) + Cu2+ → Zn2+ + Cu(s)
quickly goes to completion.
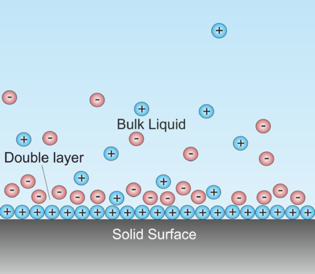
The electric double layer
[image: Larryisgood/Wikimedia]
Electrode reactions take place within the double layer, and when they do so, they alter the potential difference across this interfacial region. Much of the importance of electrochemistry lies in the fact that by applying an external voltage to the electrode, we can control both the direction and rate of an electrode reaction.
The interfacial potential differences which develop in the double layer are generally limited to only a few volts. This may not seem like very much until you consider that this potential difference spans a very small distance. In the case of an electrode immersed in a solution, this distance is essentially just the thickness of thin layer of water molecules and ions that attach themselves to the electrode surface, normally only a few atomic diameters. Thus a very small voltage can produce a very large potential gradient. For example, a potential difference of one volt across a typical 10–8 cm interfacial boundary amounts to a potential gradient of 100 million volts per centimeter— a very significant value indeed!
Interfacial potentials are not confined to metallic electrodes immersed in solutions; they can in fact exist between any two phases in contact, even in the absence of chemical reactions. In many forms of matter, they are the result of adsorption or ordered alignment of molecules caused by non-uniform forces in the interfacial region.
Interfacial potential differences are not directly observable. The usual way of measuring a potential difference between two points is to bring the two leads of a voltmeter into contact with them. It's simple enough to touch one lead of the meter to a metallic electrode, but there is no way you can connect the other lead to the solution side of the interfacial region without introducing a second electrode with its own interfacial potential, so you would be measuring the sum of two potential differences. Thus single electrode potentials, as they are commonly known, are not directly observable.
What we can observe, and make much use of, are potential differences between pairs of electrodes in electrochemical cells. This is the topic of the next lesson in this series.
More on the electric double layer (Cambridge University)
How to make your own battery, just like Allesandro Volta