What we call "classical" physics is based on our experience of what we perceive as the "real world". Even without knowing the details of Newton's laws of motion that describe the behavior of macroscopic bodies, we have all developed an intuitive understanding of this behavior; it is a part of everyone's personal view of the world.
By extension, we tend to view atoms and molecules in much the same way, that is, simply as miniature versions of the macroscopic objects we know from everyday life. It turns out, however, that our everyday view of the macroscopic world is only a first approximation of the reality that becomes apparent at the atomic level. Many of those who first encounter this microscopic world of quantum weirdness find it so foreign to prior experience that their first reaction is to dismiss it as pure fantasy.
The fact is, however, that the observed behavior of electrons in atoms and of the interaction of light with matter can only be understood in terms of quanta. The quantum view of the world of the tiny serves as the key that unlocks even some of the most fundamental aspects of modern Chemistry. Our goal in this lesson is to introduce you to this new reality, and to provide you with a conceptual understanding of it that will make Chemistry a more meaningful part of your own personal world.
Near the end of the nineteenth century, the enormous success of the recently developed kinetic molecular theory of gases had dispelled most doubts about the atomic nature of matter; the material world was seen to consist of particles that had distinct masses and sizes, and which moved in trajectories just as definite as those of billiard balls.
In the 1890s, however, certain phenomena began to be noticed that seemed to be inconsistent with this dichotomy of particles and waves. This prompted further questions and further experiments which led eventually to the realization that classical physics, while it appears to be "the truth", is by no means the whole truth. In particular, it cannot accurately describe the behavior of objects that are extremely small or fast-moving.
Chemistry began as an entirely empirical, experimental science, dealing with the classification and properties of substances and with their transformations in chemical reactions. As this large body of facts developed into a science (one of whose functions is always to explain and correlate known facts and to predict new ones), it has become necessary to focus increasingly on the nature and behavior of individual atoms and of their own constituent parts, especially the electrons. Owing to their extremely small masses, electrons behave as quantum particles which do not obey the rules of classical physics.
Quantum theory
A useful reference on this topic: the Wikipedia site on Quantum Theory
Quantum theory can be presented simply as a set of assumptions which are developed through mathematical treatment. This is in fact the best route to take if one is to use quantum mechanics as a working tool. More than this, however, quantum theory brings with it a set of concepts that have far-reaching philosophical implications and which should be a part of the intellectual equipment of anyone who claims to have a general education in the sciences. A major objective of this unit will be to introduce you to "the quantum way of thinking" and to show how this led to a profound break with the past, and a shift in our way of viewing the world that has no parallel in Western intellectual history.
New ways of looking at radiant energy
What is light?
By the mid-1600's, the prevailing opinion, championed by René Descartes and Isaac Newton, held that light consists of a flow of tiny particles. Newton noted that light seemed to react with green plants to produce growth, and must therefore be a "substance" having atoms of its own. This "corpuscular" theory of light paralleled the atomic theory of matter that was developing at the same time. Unfortunatly, it failed to explain many of the more interesting properties of light such as refraction, polarization, and interference.
Wave theory of light
In 1678, Christiaan Huygens showed how a number of optical effects could be explained if light had a wavelike nature, and this led the French engineer and scientist Augustin-Jean Fresnel to develop an elaborate wave theory of light in 1816.
By 1818 the question of "particle or wave" had become so confused that the French Academy held a great debate intended to settle the matter once and for all. The mathematician Poisson pointed out that Fresnel's wave theory had a ridiculous consequence: the shadow cast by a circular disk should have a bright spot of light at its center, where waves arriving in phase would reinforce each other. Fresnel performed the experiment and was entirely vindicated: if the light source is sufficiently point-like (an extended source such as the sun or an ordinary lamp will not work), this diffraction effect is indeed observed.
What is heat?
In the 17th century, heat was regarded as a substance called caloric whose invisible atoms could flow from one object to another, thus explaining thermal conduction. This view of heat as a material fluid seemed to be confirmed by the observation that heat can pass through a vacuum, a phenomenon that we now call radiant heat. Isaac Newton, whose experiments with a prism in 1672 led to his famous textbook "Optiks", noted that light seemed to react with green plants to produce growth, and must therefore be a "substance" having atoms of its own. By 1800, the corpuscular (particle) theory of light was generally accepted.
And yet there were questions. Count Rumford's observation that the drill bits employed in boring cannons produced more frictional heat when they were worn and dull led to the overthrow of the caloric theory.
Rise and Fall of the Caloric Theory
Radiant heat is light
By this time it was known that radiant heat and "cold" could be focussed and transmitted by mirrors, and in 1800 William Herschel discovered that radiant heat could be sensed in the dark region just beyond the red light refracted by a prism. Light and radiant heat, which had formerly been considered separate, were now recognized as one, although the question of precisely what was doing the "waving" was something of an embarrassment.
By 1890, physicists thought they had tidied up the world into the two realms of particulate matter and of wavelike radiant energy, which by then had been shown by James Clerk Maxwell to be forms of electromagnetic energy. No sooner had all this been accomplished, than the cracks began to appear; these quickly widened into chasms, and within twenty years the entire foundations of classical physics had disintegrated; it would not be until the 1920's that anyone with a serious interest in the nature of the microscopic world would find a steady place to stand.
Cathode rays become particles
The atom was the first to go. It had been known for some time that when a high voltage is applied to two separated pieces of metal in an evacuated tube, "cathode rays" pass between them. These rays could be detected by their ability to cause certain materials to give off light, or fluoresce, and were believed to be another form of electromagnetic radiation. Then, in the 1890s, J.J. Thompson and Jean Perrin showed that cathode rays are composed of particles having a measurable mass (less than 1/1000 of that of the hydrogen atom), they carry a fixed negative electric charge, and that they come from atoms. This last conclusion went so strongly against the prevailing view of atoms as the ultimate, un-cuttable stuff of the world that Thompson only reluctantly accepted it, and having done so, quickly became the object of widespread ridicule.
Radioactivity upsets the applecart
But worse was soon to come; not only were atoms shown not to be the smallest units of matter, but the work of the Curies established that atoms are not even immutable; atoms of high atomic weight such as uranium and radium give off penetrating beams of radiation and in the process change into other elements, disintegrating through a series of stages until they turn into lead. Among the various kinds of radiation that accompany radioactive disintegration are the very same cathode rays that had been produced artificially by Thompson, and which we now know as electrons.
Radiation becomes quantized
The wave theory of radiation was also running into difficulties. Any object at a temperature above absolute zero gives off radiant energy; if the object is moderately warm, we sense this as radiant heat. As the temperature is raised, a larger proportion of shorter-wavelength radiation is given off, so that at sufficiently high temperatures the object becomes luminous. The origin of this radiation was thought to lie in the thermally-induced oscillations of the atoms within the object, and on this basis the mathematical physicist James Rayleigh had worked out a formula that related the wavelengths given off to the temperature.
Unfortunately, this formula did not work; it predicted that most of the radiation given off at any temperature would be of very short wavelength, which would place it in the ultraviolet region of the spectrum. What was most disconcerting is that no one could say why Rayleigh's formula did not work, based as it was on sound classical physics; this puzzle became known as the "scandal of the ultraviolet".
Thermal emission of radiation
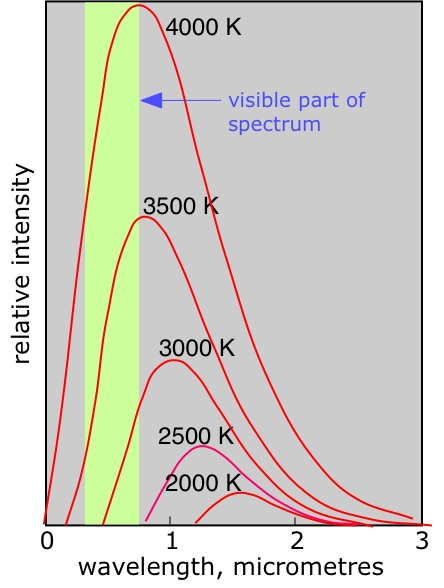
Discovering quanta
 In 1899 the German physicist Max Planck pointed out that one simple change in Rayleigh's argument would produce a formula that accurately describes the radiation spectrum of a perfect radiator, which is known as a "black body". Rayleigh assumed that such an object would absorb and emit amounts of radiation in amounts of any magnitude, ranging from minute to very large. This is just what one would expect on the basis of the similar theory of mechanical physics which had long been well established. Planck's change, for which he could offer no physical justification other than that it works, was to discard this assumption, and to require that the absorption or emission of radiation occur only in discrete chunks, or quanta. Max Planck had unlocked the door that would lead to the resurrection of the corpuscular theory of radiation. Only a few years later, Albert Einstein would kick the door open and walk through.
In 1899 the German physicist Max Planck pointed out that one simple change in Rayleigh's argument would produce a formula that accurately describes the radiation spectrum of a perfect radiator, which is known as a "black body". Rayleigh assumed that such an object would absorb and emit amounts of radiation in amounts of any magnitude, ranging from minute to very large. This is just what one would expect on the basis of the similar theory of mechanical physics which had long been well established. Planck's change, for which he could offer no physical justification other than that it works, was to discard this assumption, and to require that the absorption or emission of radiation occur only in discrete chunks, or quanta. Max Planck had unlocked the door that would lead to the resurrection of the corpuscular theory of radiation. Only a few years later, Albert Einstein would kick the door open and walk through.
The photoelectric effect
By 1900 it was known that a beam of light, falling on a piece of metal, could cause electrons to be ejected from its surface. Evidently the energy associated with the light overcomes the binding energy of the electron in the metal; any energy the light supplies in excess of this binding energy appears as kinetic energy of the emitted electron. What seemed peculiar, however, was that the energy of the ejected electrons did not depend on the intensity of the light as classical physics would predict. Instead, the energy of the photoelectrons (as they are called) varies with the color, or wavelength of the light; the higher the frequency (the shorter the wavelength), the greater the energy of the ejected electrons.
Measuring ionization energies of metallic elements
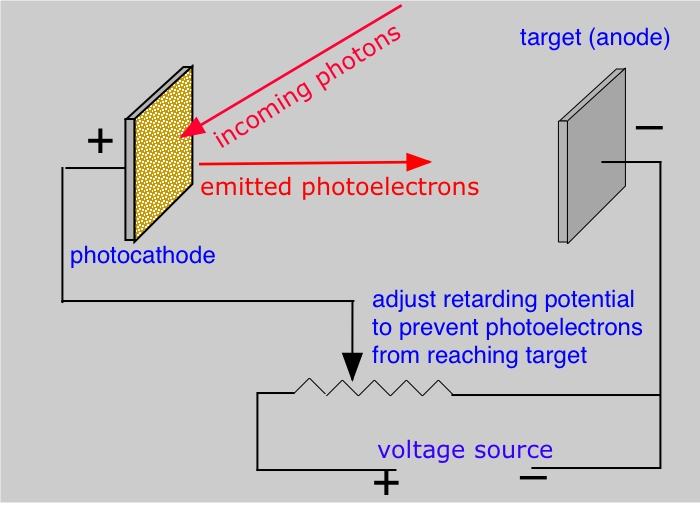
 In 1905, Albert Einstein, then an unknown clerk in the Swiss Patent Office, published a remarkable paper in which he showed that if light were regarded as a collection of individual particles, a number of phenomena, including the photoelectric effect, could be explained. Each particle of light, which we now know as a photon , has associated with it a distinct energy that is proportional to the frequency of the light, and which corresponds to Planck's energy quanta. The energy of the photon is given by
In 1905, Albert Einstein, then an unknown clerk in the Swiss Patent Office, published a remarkable paper in which he showed that if light were regarded as a collection of individual particles, a number of phenomena, including the photoelectric effect, could be explained. Each particle of light, which we now know as a photon , has associated with it a distinct energy that is proportional to the frequency of the light, and which corresponds to Planck's energy quanta. The energy of the photon is given by
![]()
in which h is Planck's constant, 6.63×10–34 J-s, ν (Greek nu) is the frequency, λ (lamda) is the wavelength, and c is the velocity of light, 3.00×108 m s–1. The photoelectric effect is only seen if the photon energy e exceeds the binding energy of the electron in the metal; it is clear from the above equation that as the wavelength increases, e decreases, and eventually no electrons will be released. Einstein had in effect revived the corpuscular theory of light, although it would not be until about 1915 that sufficient experimental evidence would be at hand to convince most of the scientific
The 1905 volume of Annalen der Physik is now an expensive collector's item, for in that year Einstein published three major papers, any one of which would have guaranteed him his place in posterity. The first, on the photoelectric effect, eventually won him the Nobel Prize. The second paper, on Brownian motion, amounted to the first direct confirmation of the atomic theory of matter. The third paper, his most famous, On the electrodynamics of moving bodies, set forth the special theory of relativity.
The appearance of his general theory of relativity in 1919 would finally make Einstein into a reluctant public celebrity and scientific superstar. This theory explained gravity as a consequence of the curvature of space-time.A neat site: Special relativity in under 15 minutes
In effect, Einstein merely asked a simple question about Faraday's law of electromagnetic induction, which says that a moving electric charge (such as is produced by an electric current flowing in a conductor) will create a magnetic field. Similarly, a moving magnetic field will induce an electric current. In either case, something has to be moving. Why, Einstein asked, does this motion have to be relative to that of the room in which the experiment is performed— that is, relative to the Earth? A stationary charge creates no field, but we know that there is really no such thing as a stationary charge, since the Earth itself is in motion; what, then, do motion and velocity ultimately relate to?
The answer, Einstein suggested, is that the only constant and unchanging velocity in the universe is that of light. This being so, the beam emitted by the headlight of a moving vehicle, for example, can travel no faster than the light coming from a stationary one. This in turn suggested (through the Lorentz transformation — we are leaving out a few steps here!) that mass, as well as velocity (and thus also, time) are relative in that they depend entirely on the motion of the observer. Two observers, moving at different velocities relative to each other, will report different masses for the same object, and will age at different rates. Further, the faster an object moves with respect to an observer, the greater is its mass, and the harder it becomes to accelerate it to a still greater velocity. As the velocity of an object approaches the speed of light, its mass approaches infinity, making it impossible for an object to move as fast as light.
![]() According to Einstein, the speed of light is really the only speed in the universe. If you are sitting still, you are moving through the time dimension at the speed of light. If you are flying in an airplane, your motion along the three cartesian dimensions subtracts from that along the fourth (time) coordinate, with the result that time, for you, passes more slowly.
According to Einstein, the speed of light is really the only speed in the universe. If you are sitting still, you are moving through the time dimension at the speed of light. If you are flying in an airplane, your motion along the three cartesian dimensions subtracts from that along the fourth (time) coordinate, with the result that time, for you, passes more slowly.
Relativity comes into chemistry in two rather indirect ways: it is responsible for the magnetic moment ("spin") of the electron, and in high-atomic weight atoms in which the electrons have especially high effective velocities, their greater [relativistic] masses cause them to be bound more tightly to the nucleus— accounting, among other things, for the color of gold, and for the unusual physical and chemical properties of mercury.
Mass and energy are united by the velocity of light
Where does the additional mass of a moving body come from? Simply from the kinetic energy of the object; this equivalence of mass and energy, expressed by the famous relation e = mc2, is the most well known consequence of special relativity. The reason that photons alone can travel at the velocity of light is that these particles possess zero rest mass to start with. You can think of ordinary matter as "congealed energy", trapped by its possession of rest mass, whereas light is energy that has been liberated of its mass.



