Chemical energetics
An introduction to chemical thermodynamics
On this page:
Energy is one of the most fundamental and universal concepts of physical science, but one that is remarkably difficult to define in way that is meaningful to most people. This perhaps reflects the fact that energy is not a “thing” that exists by itself, but is rather an attribute of matter (and also of electromagnetic radiation) that can manifest itself in different ways. It can be observed and measured only indirectly through its effects on matter that acquires, loses, or possesses it.
The concept that we call energy was very slow to develop; it took more than a hundred years just to get people to agree on the definitions of many of the terms we use to describe energy and the interconversion between its various forms. But even now, most people have some difficulty in explaining what it is; somehow, the definition we all learned in elementary science ("the capacity to do work") seems less than adequate to convey its meaning.
Although the term "energy" was not used in science prior to 1802, it had long been suggested that certain properties related to the motions of objects exhibit an endurance which is incorporated into the modern concept of "conservation of energy". René Descartes (1596-1650) stated it explicitly:
When God created the world, He "caused some of its parts to push others and to transfer their motions to others..." and thus "He conserves motion".*
In the 17th Century, the great mathematician Gottfried Leibniz (1646-1716) suggested the distinction between vis viva ("live force") and vis mortua ("dead force"), which later became known as kinetic energy (1829) and potential energy (1853).
Kinetic energy and potential energy
Whatever energy may be, there are basically two kinds.
Kinetic energy is associated with the motion of an object, and its direct consequences are part of everyone's daily experience; the faster the ball you catch in your hand, and the heavier it is, the more you feel it. Quantitatively, a body with a mass m and moving at a velocity v possesses the kinetic energy mv2/2.
Potential energy is energy a body has by virtue of its location. But there is more: the body must be subject to a "restoring force" of some kind that tends to move it to a location of lower potential energy. Think of an arrow that is subjected to the force from a stretched bowstring; the more tightly the arrow is pulled back against the string, the more potential energy it has.
More generally, the restoring force comes from what we call a force field— a gravitational, electrostaticl, or magnetic field. We observe the consequences of gravitational potential energy all the time, such as when we walk, but seldom give it any thought.
If an object of mass m is raised off the floor to a height h, its potential energy increases by mgh, where g is a proportionality constant known as the acceleration of gravity; its value at the earth's surface is 9.8 m s–2.
Similarly, the potential energy of a particle having an electric charge q depends on its location in an electrostatic field.
"Chemical energy"
Electrostatic potential energy plays a major role in chemistry; the potential energies of electrons in the force field created by atomic nuclei lie at the heart of the chemical behavior of atoms and molecules.
"Chemical energy" usually refers to the energy that is stored in the chemical bonds of molecules. These bonds form when electrons are able to respond to the force fields created by two or more atomic nuclei, so they can be regarded as manifestations of electrostatic potential energy.
In an exothermic chemical reaction, the electrons and nuclei within the reactants undergo rearrangment into products possessing lower energies, and the difference is released to the environment in the form of heat.
Interconversion of potential and kinetic energy
Transitions between potential and kinetic energy are such an intimate part of our daily lives that we hardly give them a thought. It happens in walking as the body moves up and down.

Our bodies utilize the chemical energy in glucose to keep us warm and to move our muscles. In fact, life itself depends on the conversion of chemical energy to other forms.
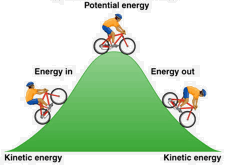 Energy is conserved: it can neither be created nor destroyed. So when you go uphill, your kinetic energy is transformed into potential energy, which gets changed back into kinetic energy as you coast down the other side. And where did the kinetic energy you expended in peddling uphill come from? By conversion of some of the chemical potential energy in your breakfast cereal.
Energy is conserved: it can neither be created nor destroyed. So when you go uphill, your kinetic energy is transformed into potential energy, which gets changed back into kinetic energy as you coast down the other side. And where did the kinetic energy you expended in peddling uphill come from? By conversion of some of the chemical potential energy in your breakfast cereal.
- When you drop a book onto the floor, its potential energy is transformed into kinetic energy. When it strikes the floor, this transformation is complete. What happens to the energy then? The kinetic energy that at the moment of impact was formerly situated exclusively in the moving book, now becomes shared between the book and the floor, and in the form of randomized thermal motions of the molecular units of which they are made; we can observe this effect as a rise in temperature.
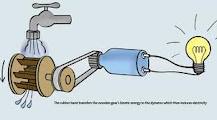
- ← Much of the potential energy of falling water can be captured by a water wheel or other device that transforms the kinetic energy of the exit water into kinetic energy. The output of a hydroelectric power is directly proportional to its height above the level of the generator turbines in the valley below. At this point, the kinetic energy of the exit water is transferred to that of the turbine, most of which (up to 90 percent in the largest installations) is then converted into electrical energy.
- Will the temperature of the water at the bottom of a water fall be greater than that at the top? James Joule himself predicted that it would be. It has been calculated that at Niagra falls, that complete conversion of the potential energy of 1 kg of water at the top into kinetic energy when it hits the plunge pool 58 meters below will result in a temperature increase of about 0.14 C°. (But there are lots of complications. For example, some of the water breaks up into tiny droplets as it falls, and water evaporates from droplets quite rapidly, producing a cooling effect.)
 Chemical energy can also be converted, at least partially, into electrical energy: this is what happens in a battery. If a highly exothermic reaction also produces gaseous products, the latter may expand so rapidly that the result is an explosion — a net conversion of chemical energy into kinetic energy (including sound).
Chemical energy can also be converted, at least partially, into electrical energy: this is what happens in a battery. If a highly exothermic reaction also produces gaseous products, the latter may expand so rapidly that the result is an explosion — a net conversion of chemical energy into kinetic energy (including sound).
Thermal energy
Kinetic energy is associated with motion, but in two different ways. For a macroscopic object such as a book or a ball, or a parcel of flowing water, it is simply given by ½ mv2.
But as we mentioned above, when an object is dropped onto the floor, or when an exothermic chemical reaction heats surrounding matter, the kinetic energy gets dispersed into the molecular units in the environment. This "microscopic" form of kinetic energy, unlike that of a speeding bullet, is completely random in the kinds of motions it exhibits and in its direction. We refer to this as "thermalized" kinetic energy, or more commonly simply as thermal energy. We observe the effects of this as a rise in the temperature of the surroundings. The temperature of a body is direct measure of the quantity of thermal energy is contains.
Convsersion of thermal energy to heat is never completely recoverable
Once kinetic energy is thermalized, only a portion of it can be converted back into potential energy. The remainder simply gets dispersed and diluted into the environment, and is effectively lost.
To summarize, then:
- Potential energy can be converted entirely into kinetic energy..
- Potential energy can also be converted, with varying degrees of efficiency,into electrical energy.
- The kinetic energy of macroscopic objects can be transferred between objects (barring the effects of friction).
- Once kinetic energy becomes thermalized, only a portion of it can be converted back into either potential energy or be concentrated back into the kinetic energy of a macroscopic. This limitation, which has nothing to do with technology but is a fundamental property of nature, is the subject of the second law of thermodynamics.
- A device that is intended to accomplish the partial transformation of thermal energy into organized kinetic energy is known as a heat engine.








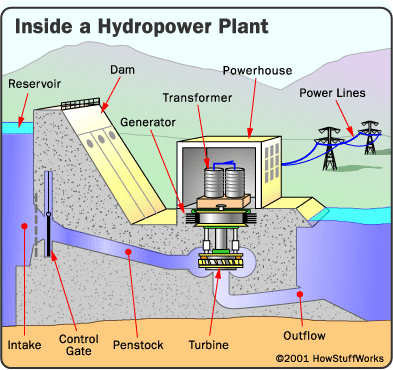 Schematic diagram of a
Schematic diagram of a Turbine being installed at Grand Coulee dam, 1974.
Turbine being installed at Grand Coulee dam, 1974. 
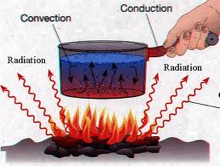 Transfer of thermal energy can be accomplished by bringing two bodies into physical contact (the kettle on top of the stove, or through an electric heating element inside the kettle). Another mechanism of thermal energy transfer is by radiation; a hot object will convey energy to any body in sight of it via electromagnetic radiation in the infrared part of the spectrum. In many cases, a combination of modes will be active:
Transfer of thermal energy can be accomplished by bringing two bodies into physical contact (the kettle on top of the stove, or through an electric heating element inside the kettle). Another mechanism of thermal energy transfer is by radiation; a hot object will convey energy to any body in sight of it via electromagnetic radiation in the infrared part of the spectrum. In many cases, a combination of modes will be active:
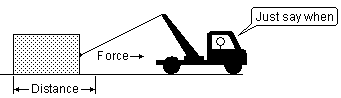 Work, like energy, can take various forms, the most familiar being mechanical and electrical. Mechanical work arises when an object moves a distance Δx against an opposing force f: w = f Δx N-m; 1 N-m = 1 J.
Work, like energy, can take various forms, the most familiar being mechanical and electrical. Mechanical work arises when an object moves a distance Δx against an opposing force f: w = f Δx N-m; 1 N-m = 1 J.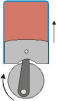 Work can be completely converted into heat (by friction, for example), but heat can only be partially converted to work. Conversion of heat into work is accomplished by means of a heat engine, the most common example of which is an ordinary gasoline engine. The science of
Work can be completely converted into heat (by friction, for example), but heat can only be partially converted to work. Conversion of heat into work is accomplished by means of a heat engine, the most common example of which is an ordinary gasoline engine. The science of 