
chem1 virtual textbook
a reference text for General Chemistry
Stephen Lower
Simon Fraser University
The Chem1 Virtual Textbook is a
resource for General Chemistry aimed mainly at the first-year
university level.
It offers a more comprehensive, organized,
and measured approach than is found in most standard
textbooks. It should also be accessible to students in advanced
high-school courses, and helpful as review material for
students in more advanced courses in chemistry, biology,
geology, and engineering.
1- Introduction to acid-base equilibria
- Fundamentals of proton-transfer equilibria
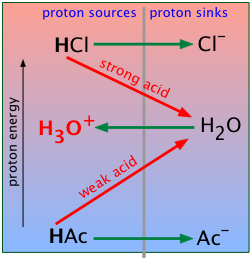
- The fall of the proton and the proton-free energy scale
- Why acids react with bases, but not always completely
- What you should be able to do
- Appendix: pH, pKa and the average proton free energy
2- Strong monoprotic acids and bases
- At moderate concentrations, forget about equilibria!
- Getting to know the major strong acids and bases
- Acids behave unpredictably at high concentrations
- Systematic calculations for very dilute solutions
- What you should be able to do
3- Finding the pH of weak acids and bases, and salts
- Understanding Ca, [HA], and degree of dissociation
- Details of pH calculations; dealing with (or avoiding) quadratic equations
- Solutions of polyprotic acids; simplifying approximations; sulfuric acid
- Amino acids and Zwitterions
- What you should be able to do
4- Mixtures of conjugate pairs; Buffer solutions
- How the pH controls conjugate pair concentration ratios
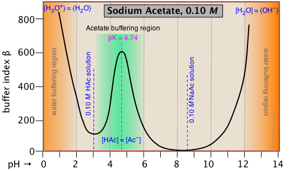
- Buffer solutions and calculations; the Henderson-Hasselbalch equation
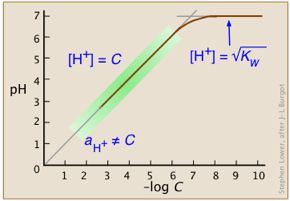
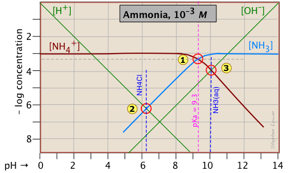
- Calculations without algebra; log-C vs pH (Sillén) plots
- How to make and use Sillén diagrams
- What you should be able to do
- Understanding titration curves
- Detecting the equivalence point
- Titration-related calculations
- What you should be able to do
6- Some applications of acid-base equilibria
- Buffers are everywhere!
- Acid-base chemistry in physiology and medicine
- Acid rain
- The geochemical carbonate system
- What you should be able to do
7- Exact calculations and their approximations
These topics are not ordinarily covered in General Chemistry courses, but are often needed for more advanced courses in analytical-, environmental-, and aquatic chemistry.
