 Fig 6 - Solution of ammonium acetate in water
Fig 6 - Solution of ammonium acetate in water
 Fig 6 - Solution of ammonium acetate in water
Fig 6 - Solution of ammonium acetate in water
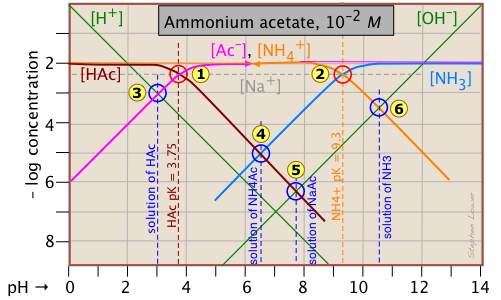
The quantitative treatment of a solution of a salt of a weak acid and a weak base is algebraically complicated, even when done to the crudest approximation. Graphically, it is a piece of cake! As you can see above, we just construct plots of the two acid-base system on the same graph. The stoichiometry of the salt defines the system point at about pH 6.5, showing that hydrolyses of the two systems does not quite "cancel out". Of course, this is still an approximation that neglects ioniziation of either the anion or cation, but it is probably as valid as any calculation that is likely to be carried out ordinarily-available data.