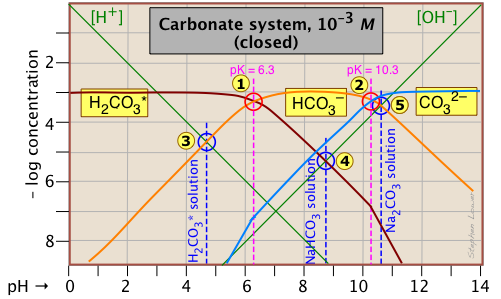

Here it is — the most important of all acid-base systems in natural waters and physiology! Nothing new as far as theory goes-- see the explanation given for oxalic acid. What's different heee is the two sts of plots. The lower (more dilute) one corresponds to the concentration of dissolved CO2 present in water in equlibrium with the atmosphere having the normal CO2 partial pressure of about .0005 atm. You can see from this that when pure water is exposed to the atmosphere, its pH will fall from 7 to about 5 (but see the last paragraph below); thus all rain is in a sense "acid rain".
Note that around pH 7.4 - 7.8 (the pH of blood and of the ocean), over 99% of all carbonate is in the form of bicarbonate HCO3–. As a result, the ocean acts as a sink for atmospheric CO2 and contains about 50 times as much as does the atmosphere. Similarly, CO2 resulting from cellular respiration is converted to HCO3– for transport through the blood stream, and is converted back to CO2 in the lungs.
This plot for the 10–3 M system corresponds to the approximate carbonate content of natural waters in contact with rocks and sediments (which normally contain carbonate components). It describes the situation in lakes, streams, ground waters and the ocean. To work this out for a 10–5 M system that corresponds to a solution in equilibrium with CO2 at it partial pressure in the atmosphere, just translate all the carbonate plots down by 2 log units.
The higher the partial pressure of CO2 , the lower will be the pH of a carbonate-containing system. This is the reason the pH of an algae-containing pond rises during the day (when CO2 is being consumed by photosynthesis) and falls at night (CO2 restored by respiration.) More seriously, this explains why human-generated increases in atmospheric CO2 is driving down the pH of the oceans, gradually destroying the viability of marine organism from corals to shellfish.
The term "closed system" in the caption at the top of the graph means that CT, the sum of the concentrations of all three carbonate species, is assumed constant here. For an open system the plots for the bicarbonate and carbonate concentrations would be dramatically different because an alkaline solution open to the atmosphere has access to an infinite supply of CO2 and will absorb it up to the colubility limit of sodium carbonate. (You may recall that a common test for CO2 is to watch for a precipitate to appear in a saturated solution of calcium hydroxide.) Thus the estimate of pH=5 for pure water exposed to the atmosphere is not quite correct; using an "opon system" plot, a somewhat lower pH, around 4.5, will be found. It turns out, however, that for many practical situations, the relatively slow transport of CO2 between air and water makes the "closed system" predictions reasonably accurate within the time scale of interest.
Some related pages of interest